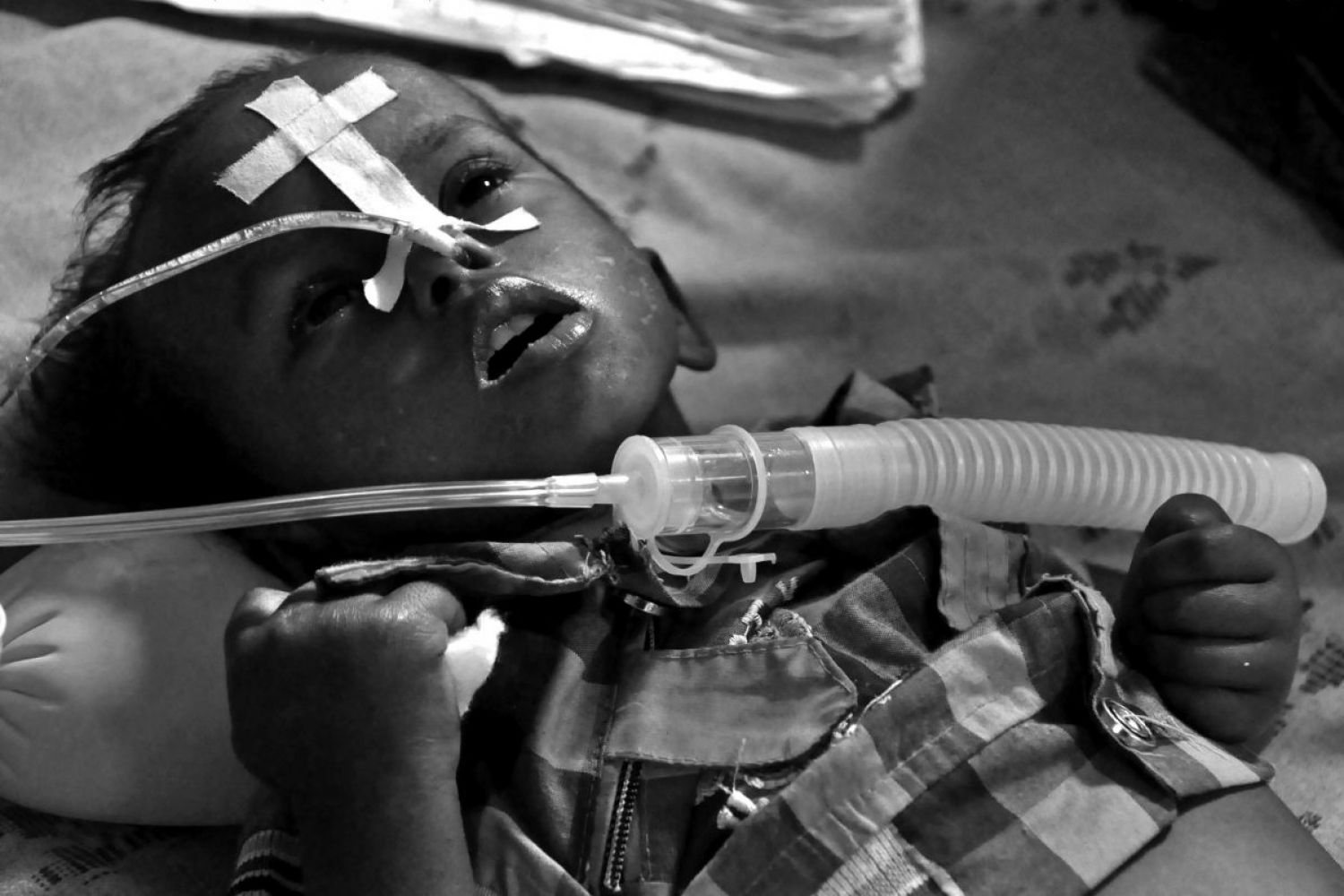
Each year as
monsoon-bearing clouds arrive over eastern Uttar Pradesh, so does a pestilence.
Hundreds, mostly children, fall ill with fever, convulsions, and
disorientation. So familiar are people with these symptoms that at the first
signs they rush their children to Baba Raghav Das Medical College Hospital,
Gorakhpur. Few other hospitals in the eastern Uttar Pradesh region either have
the doctors or the equipment to handle this affliction. And so, the journey of
children from nearly 15 d





